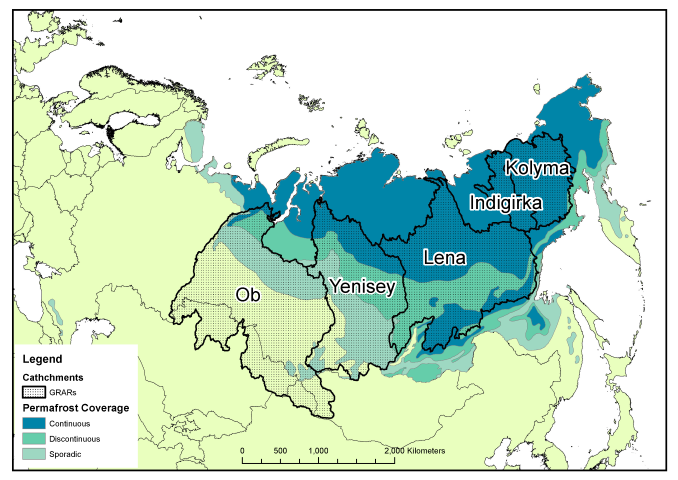
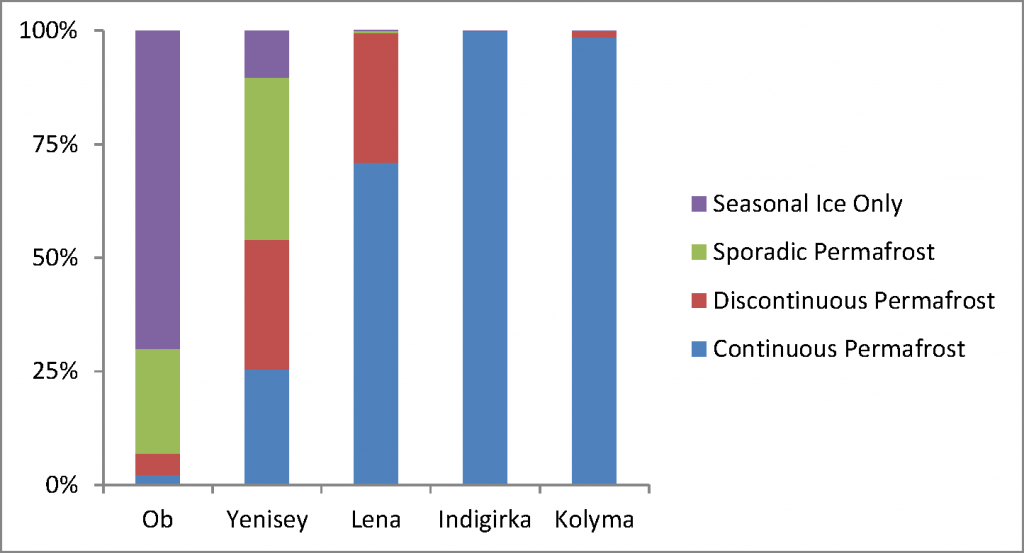
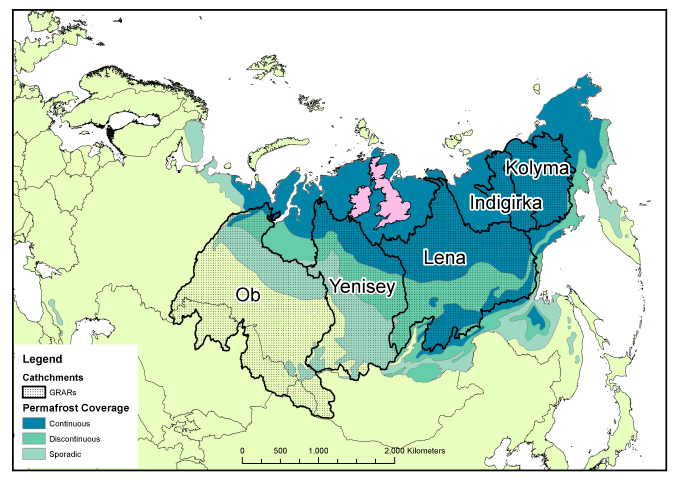
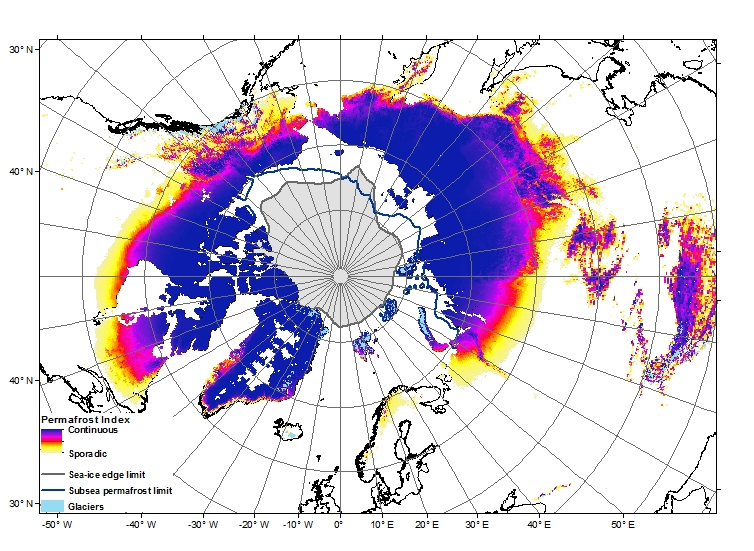
Peer review is generally accepted to be the least-worst way to generate trust in the scientific publishing process. By allowing experts in the field to read, critique, confirm, challenge and improve your work before it enters the mainstream body of science, poor quality or erroneous work should be filtered out before it gets the chance to distort public perception and policy. However, it’s not without its critics. Anonymous reviews allow reviewers to partake in spiteful and/or personal attacks which do nothing to improve the science behind the work, and can delay or even prevent publication of perfectly acceptable work. Also, the system is based on a financial system that only seems to benefit the publishing companies. In traditional journals, scientists relinquish their copyright to a company that then charges them and their colleagues to read the work, restricting access to those in universities or with big budgets (individual papers can cost $30 or more, subscriptions run to the thousands). Journal reviewers and editors work for free, considering the process as part of their community obligation despite the for-profit journal getting the real benefits.
Recently, open-access publishing has started to change the way that ordinary people can read the research that they, through their taxes and charity donations, have paid for, but the business model for the publishers is generally similar. In a typical open access publishing workflow, the researchers submitting the paper pay an “article processing charge” (APC) once the work has been accepted. Paying this charge, which is often £1000 or more, allows them to retain the copyright, and lets anyone, anywhere in the world, read the paper for free. It shifts the costs for access from the distributed consumers, who would often lack the resources to pay for the research, to the universities producing the work in the first place. Most research grant bodies now request open access publishing, and have provided some funds to cover the APCs, for now at least. Reviewers and editors are still unpaid, and publishers still make a profit, in fact since many articles are not open access then universities (i.e. taxpayers) are on the hook for both the journal subscriptions and the APCs.
So why are researchers still paying these companies such large amounts of money (profit margins are amongst the best of any industry)? Well, academic promotion is mostly decided by your publication history; the easiest way to judge a publication history is to look at the journals that a researcher publishes in, rather than reading the papers themselves. Therefore the pressure is on, especially for young researchers, to publish in the most prestigious journals, and they tend to be the most expensive ones, where articles are either restricted access or have high APCs.
Recently, there has been a shift towards more open and accountable publishing systems, with journals allowing researchers to publish either draft versions or even the finished paper on their website without violating copyright. Many universities have created online repositories to let researchers store and share their work (mine is available through my Manchester profile page), which are imperfect, since it’s often hard to find the papers, but better than nothing.
Even within my short career, the way that people publish and access science has changed; open publishing is still in development and it’s likely that further innovation in the next 5-10 years will change the landscape even further.